Bilthoven Biologicals and Tonix Pharmaceuticals collaborate on viral vector-based Mpox vaccine.
Bilthoven Biologicals and Tonix Pharmaceuticals are collaborating to develop TNX‑801, a viral vector-based Mpox vaccine aimed at enhancing global vaccine availability.
Bilthoven Biologicals and Tonix Pharmaceuticals enter collaboration to advance development of viral vector-based Mpox vaccine (formerly known as monkeypox vaccine) TNX-801
On August 14, 2024, the WHO declared monkeypox a Public Health Emergency of International Concern (PHEIC) for the second time in two years.
Mpox is endemic in parts of Africa.
Global availability and affordability of an Mpox vaccine are essential to improving worldwide health.
The latest Clade 1 strain poses a known global threat with a mortality rate up to 10%.
Clinical research program led by Tonix, manufacturing led by Bilthoven Biologicals.
Bilthoven Biologicals (“BBio”), part of the Cyrus Poonawalla Group, and Tonix Pharmaceuticals (“Tonix”) announced a partnership to further develop their Mpox vaccine candidate (“TNX-801”). Tonix is a fully integrated biopharmaceutical company with marketed products and a pipeline of development candidates. BBio is a global vaccine company producing both prophylactic and pediatric vaccines as well as therapeutic vaccines. BBio was selected by the EU for its pandemic preparedness program of “always-warm” vaccine manufacturing facilities.
Both companies are committed to the development and commercialization of the Mpox vaccine candidate. TNX-801 is based on viral vector technology, with the potential to serve as a viral vector platform allowing recombinant versions to be developed to protect against other infectious diseases.
About TNX-801
TNX-801 (recombinant horsepox virus) is a live attenuated horsepox-based vaccine currently in preclinical development to prevent chickenpox and smallpox. Tonix reported positive preclinical efficacy data showing vaccination with TNX-801 protected non-human primates from lethal monkeypox exposure. Tonix received formal written feedback from a Type B pre-Investigational New Drug (IND) meeting with the U.S. Food and Drug Administration (FDA) regarding the development of TNX-801 as a potential vaccine for protection against chickenpox and smallpox. Tonix believes FDA feedback provides a pathway toward agreement on the design of a Phase 1/2 study and overall clinical development plan. More than 90,000 people worldwide contracted monkeypox during the 2022-23 outbreak. The June 2023 monkeypox cluster in Chicago revealed breakthrough cases in individuals vaccinated with the currently approved non-replicating two-dose vaccine. TNX-801, by contrast, is administered percutaneously in a single dose, potentially achieving higher community protection rates by eliminating dropouts between doses and reducing forward transmission. Moreover, reliance on only one approved monkeypox vaccine currently presents a risk to the global supply chain, which has already led to vaccine shortages worldwide, particularly in Africa. TNX-801 has the potential to make a global impact on monkeypox risk due to its durable T-cell immune response, scalable manufacturing, and lower dose requirements compared to non-replicating vaccines.
Seth Lederman, CEO of Tonix, said: “We look forward to working with BBio to accelerate the development of our candidate vaccines to prevent smallpox, monkeypox, and other infectious diseases. TNX-801 is delivered in a single dose, which we believe will improve uptake and eliminate partial vaccination compared to current two-dose regimens. We believe TNX-801 can be rapidly scaled for production and distributed and stored without the need for expensive and complex ultra-cold supply chains.”
Pandemic potential of Mpox
The global Mpox outbreak starting in 2022 affected over 90,000 individuals in countries where Mpox was previously non-endemic, including Europe and the U.S. The spread of Clade IIb Mpox in 2022 highlights the pandemic potential of the disease. Mpox is currently endemic in several Central African countries, including the Democratic Republic of Congo, where the highly lethal Clade 1 strain causes mortality rates up to 10%.
“The recent Mpox outbreak perfectly illustrates why we built the Pandemic Preparedness Facility at BBio,” said BBio CEO Jurgen Kwik. “Establishing the ‘ever-warm’ pandemic preparedness facility underscores the critical importance of readiness for global health crises like Mpox. This collaboration highlights the essential role of the facility in strengthening pandemic preparedness and response.”
Global availability of an affordable, safe, and effective single-dose Mpox vaccine is crucial given the disease’s pandemic potential. Successful development of TNX-801 will lay the foundation for expanding the viral vector platform, enabling recombinant versions to protect against other infectious diseases and future outbreaks.
Jorrit van Hoorn, Director Corporate Development at Bilthoven Biologicals, added: “We look forward to this groundbreaking collaboration with Tonix and contributing to a solution for this global health emergency, leveraging the pandemic facilities at BBio together with the world-leading vaccine expertise of our parent company, the Serum Institute of India.”
The full press release and more information about Tonix can be found at www.tonixpharma.com.
1Noyce RS, et al. Viruses. 2023;15(2):356. DOI: https://doi.org/10.3390/v15020356
2TNX-801 PR pre-IND meeting 8/20/23: https://ir.tonixpharma.com/news-events/press-releases/detail/1417/tonix-pharmaceuticals-announces-results-of-pre-ind-meeting
3CDC. (2022-2023). Mpox Outbreak Global Map https://www.cdc.gov/poxvirus/mpox/response/2022/world-map.html
4 Faherty EA, et al. MMWR Morb Mortal Wkly Rep. 2023;72:696–698. DOI: http://dx.doi.org/10.15585/mmwr.mm7225a6.
Bron: Bilthoven Biologicals
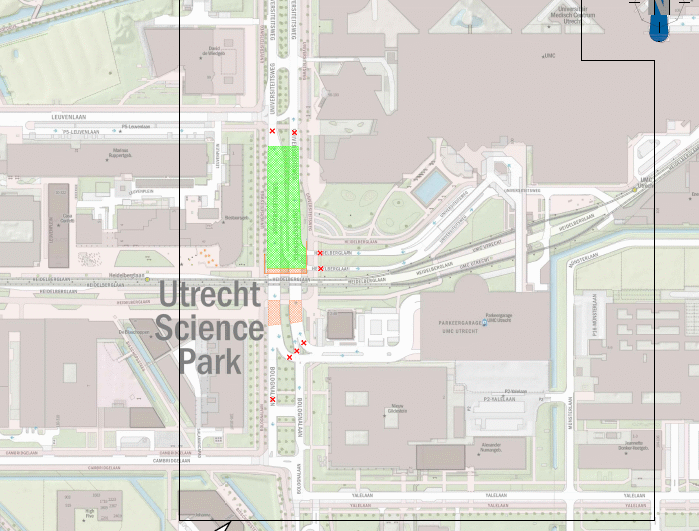
Roadworks Universiteitsweg (phase 2) from 24 to 26 April
In the weekend from Friday 24 April to Sunday 26 April, the second phase of the works on Universiteitsweg will be carried out. This phase involves replacing the asphalt surface layers on the southern section of the road, between Leuvenlaan and the tram tracks.

Looking back at the successful Utrecht Science Park Spring Café: the power of an ecosystem in setting up and running a startup
On Thursday, April 9, the Utrecht Science Park Spring Café took place at UtrechtInc. In an informal setting, colleagues, partners, and stakeholders gathered to meet and catch up on developments within the Utrecht Science Park.

Utrecht Science Week: Art and science unite at the Betweter Festival on 25 September
From 25 September to 4 October, the Utrecht Science Park will be alive with curiosity and exploration during the annual Utrecht Science Week. For over a week, the Utrecht Science Park opens its doors to everyone interested in both applied and fundamental science. In close collaboration with various partners, a diverse programme is put together, with special focus on themes such as sustainability and health.

Inspiring and connecting visit by the President of Estonia to Utrecht Science Park
On Wednesday, April 8, 2026, the President of Estonia, Mr. Alar Karis, together with his wife Sirje Karis, ambassador Mr. Paul Teesalu and a delegation from the Estonian Health Ecosystem, visited the Utrecht Science Park. During the visit, much knowledge was exchanged and valuable connections were made.